- Home
- The Arizona News
- FDA Approves First Systemic Treatment for Alopecia Areata
FDA Approves First Systemic Treatment for Alopecia Areata
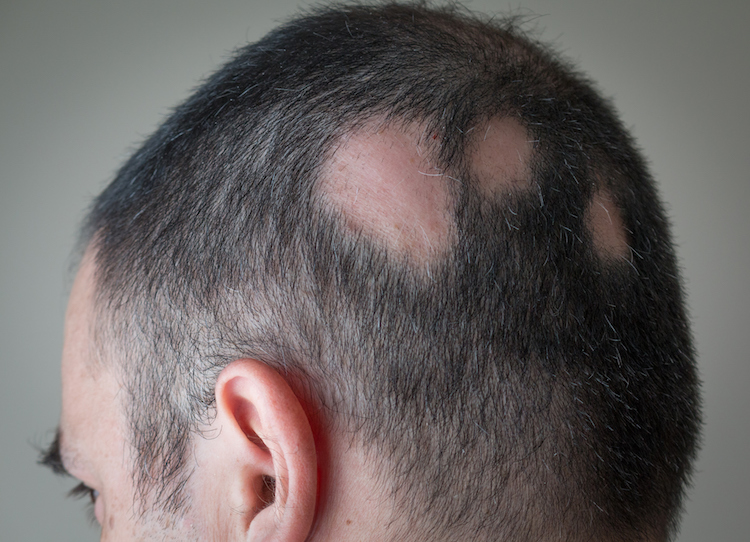
This week, the U.S. Food and Drug Administration approved Olumiant (baricitinib) oral tablets to treat adult patients with severe alopecia areata, a disorder that often appears as patchy baldness and affects more than 300,000 people in the U.S. each year. Today’s action marks the first FDA approval of a systemic treatment, which treats the entire body rather than a specific location for alopecia areata.
“Access to safe and effective treatment options is crucial for the significant number of Americans affected by severe alopecia,” said Kendall Marcus, M.D., director of the Division of Dermatology and Dentistry in the FDA’s Center for Drug Evaluation and Research. “Today’s approval will help fulfill a significant unmet need for patients with severe alopecia areata.”
Alopecia areata, commonly referred to as just alopecia, is an autoimmune disorder in which the body attacks its own hair follicles, causing hair to fall out, often in clumps. Olumiant is a Janus kinase (JAK) inhibitor which blocks the activity of one or more of a specific family of enzymes, interfering with the pathway that leads to inflammation.
The efficacy and safety of Olumiant in alopecia areata was studied in two randomized, double-blind, placebo-controlled trials Trial AA-1 and Trial AA-2 with patients who had at least 50% scalp hair loss as measured by the Severity of Alopecia Tool for more than six months. Patients in these trials received either a placebo, 2 milligrams of Olumiant, or 4 milligrams of Olumiant every day. The primary measurement of efficacy for both trials was the proportion of patients who achieved at least 80% scalp hair coverage at week 36.
In Trial AA-1, 22% of the 184 patients who received 2 milligrams of Olumiant and 35% of the 281 patients who received 4 milligrams of Olumiant achieved adequate scalp hair coverage, compared to 5% of the 189 patients who received a placebo. In Trial AA-2, 17% of the 156 patients who received 2 milligrams of Olumiant and 32% of the 234 patients who received 4 milligrams of Olumiant achieved adequate scalp hair coverage, compared to 3% of the 156 patients who received a placebo.
The most common side effects associated with Olumiant include: upper respiratory tract infections, headache, acne, high cholesterol (hyperlipidemia), increase of an enzyme called creatinine phosphokinase, urinary tract infection, liver enzyme elevations, inflammation of hair follicles (folliculitis), fatigue, lower respiratory tract infections, nausea, genital yeast infections (Candida infections), anemia, low number of certain types of white blood cells (neutropenia), abdominal pain, shingles (herpes zoster) and weight increase.
Olumiant was originally approved in 2018. It is approved as a treatment for certain adult patients with moderately to severely active rheumatoid arthritis. Olumiant is also approved for the treatment of COVID-19 in certain hospitalized adults.
